Contrary to what many viewers may have expected, atomic models and experiments involving their of have been assigned this page do to the contributors having ample enjoyment with information. Do not worry, each have been set in proper order.
Changes to Atomic Theory and Model (experiments are "*")
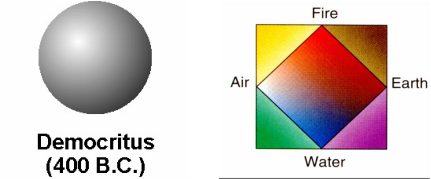
Democritus vs. Aristotle No, this is not a Greek equivalent of a baseball. It is what Democritus envisioned an atom would be. He suggested that atoms were small invisible particles that are indivisible by nature. With no experimental data to back up this model, it was later replaced by Aristotle's elemental proposal which at that time seemed much more appropriate. As many should know by now this interpretation of the universe's "building blocks" did not change until centuries later.
|
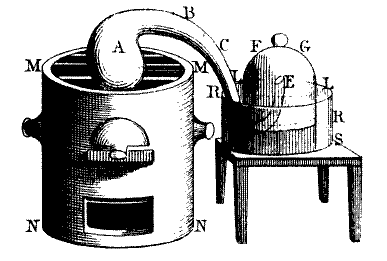
Law of Conservation of Mass Although he had little to do with the atomic theory, Antoine Lavoisier's experiment did contribute a great portion to chemistry. The Law of Conservation of Mass was created through his experiment involving melted metal. His experiment showed that the liquid metal gained mass as the air around lost it.
|
Law of Constant Composition Stating that the ratio in compounds are always the same, the Law of Constant Composition made life easier to chemists alike by proving that elements combined in chemical compounds that are separated will have virtually the same mass ratio as they would when they were together. So if separated the mass proportions will always be the same.
|
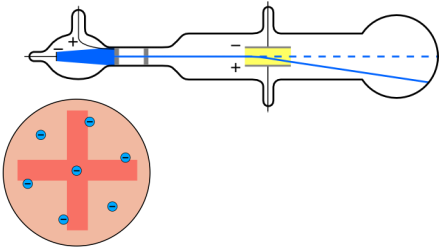
The Electron/Plum Pudding By attaching two magnets into the inside of his cathode ray, J.J Thomson detected the change in direction of the ray whenever he flipped the positively charged and negatively charged magnets. The cathode rays always went towards the positive charge, causing J.J Thomson to create a model that contained a negatively charged particle, electrons. He suspected that electrons were floating inside a positively charged material.
|
The Proton By discovering the nucleus of an atom, Ernest Rutherford also unintentionally discovered the proton. Through his gold foil experiment, where he shot an alpha particle beam at gold foil surrounded by zinc sulfide. His data showed that the particles bounced around inside the atom. From this he created a model that showed an atom with a compacted area in the middle called a nucleus where positive charges were held.
|
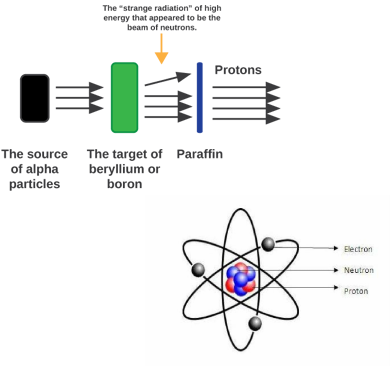
The Neutron When researching the radiation atoms omit, James Chadwick discovered a neutral charge inside the atom's nucleus. By trying to find a proton electron pair in his experiment of blasting alpha particles at Beryllium, Chadwick identified that when they pass through a magnetic field, they weren't being deflected. With this in mind, he concluded that this was the Neutron.
|
The Current Model/Theory
|
Dalton's Atomic Theory explanation did not appear until now because the theory still pertained basic principals that are used today:
1. All matter is composed of microscopic particles known as atoms. The nucleus contains neutrons (neutral) and protons (positive) with an electron cloud filled with, use your best guess. 2. Atoms are not created nor can they be destroyed in nature. 3. Atoms in an element are all identical. 4. Atoms of uncommon elements have different chemical properties and weights. (mass is not always equal in atoms of the same element) 5. Atoms of different elements can combine to form compounds. However, there were some flaws so the theory now exists as: All matter is constructed from microscopic indivisible particles called atoms. Atoms of a certain element are nearly identical, and differ from other elements. Atoms also combine to form compounds with fixed proportions. |